Geneos is currently pursuing clinical development to treat cancer patients with personalized neoantigen-directed immunotherapy.
1
Hepatocellular Carcinoma
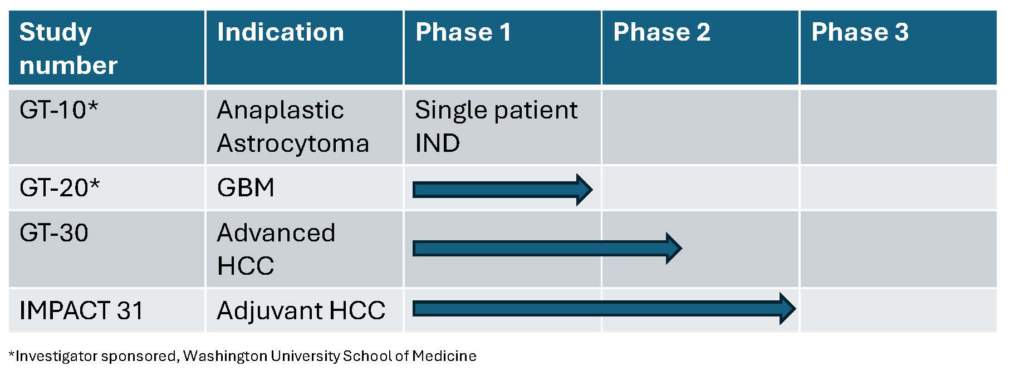
- GT-30: a Geneos-sponsored Phase 1b/2a clinical trial to treat patients with advanced hepatocellular carcinoma (HCC). Click here for more information about the clinical trial.
- GT-31: Phase 2b randomized, controlled clinical trial as adjuvant immunotherapy in patients with HCC.
2
GBM/Anaplastic Astrocytoma
- GT-10: Single patient compassionate use of our neoantigen-targeted immunotherapy for the treatment of a patient with anaplastic astrocytoma.
- GT-20: an investigator-sponsored Phase I clinical trial in collaboration with Washington University to treat patients with newly diagnosed glioblastoma multiforme (GBM). Click here for more information about the clinical trial.
About GBM
Glioblastoma (GBM) is the most common primary malignant brain tumor accounting for approximately 13,000 new cases per year. GBM also carries the worst prognosis of primary gliomas with a dismal median survival of 12-15 months despite aggressive combination treatment. Current standard of care therapy involves post-resection concurrent radiotherapy plus temozolomide followed by six months of maintenance temozolomide. Unfortunately, the vast majority of patients will eventually relapse, and since there is a relative paucity of proven effective second-line options available, patients rapidly succumb to their disease with the current 5-year survival rate <10%. Thus, new therapeutic modalities are desperately needed. On GT-20, Geneos is testing GNOS-PV01 (our personalized neoantigen-targeted immunotherapy delivered intramuscularly) in combination with IL-12 adjuvant for the treatment of patients with newly diagnosed GBM following surgery and radiation.
About HCC
Hepatocellular carcinoma (HCC) is the sixth most common cancer worldwide with over 700,000 annual diagnoses and is the fourth most common cause of cancer-related death worldwide. The incidence of HCC is rising in US and Europe. In most patients, HCC arises in conjunction with liver cirrhosis and is attributed to several risk factors, including infection (usually with hepatitis B or C viruses), alcohol consumption, and non- alcoholic fatty liver disease. Most HCC patients present with advanced disease. These patients are typically treated worldwide with systemic therapies. Today, Tyrosine Kinase Inhibitors (TKI) and checkpoint inhibitors are indicated for the treatment of advanced HCC. However, in most patients, responses to these therapies last between 3-4 months, and the 5-year survival rate of HCC is 18.4%. The Geneos GT-30 study has investigated the use of GNOS-PV02 (our personalized neoantigen-targeted immunotherapy delivered intradermally) in combination with IL-12 adjuvant and anti-PD1 for the treatment of patients with advanced HCC, with patients switched to GNOS-PV02 monotherapy either after two years or sooner in the case of immune toxicity from anti-PD1.